
In retrospect, 2016 was a banner year for exploding devices. The trend began when hoverboards — two-wheeled self-balancing motorized devices — randomly began to burst into flames, hurting riders and causing millions of dollars in property damages. Samsung’s Galaxy Note 7 mobile phone, which debuted to stellar reviews in August, faced similar issues and had to be withdrawn from the market by October. It turns out that all the mishaps were the result of malfunctioning lithium-ion (Li-ion) batteries used to power the devices.
This, of course, is not the first time devices using lithium-ion batteries have had issues. In 2006, Dell Computers recalled 4.1 million notebook computer batteries because they could erupt in flames. In 2007, Nokia did the same for 46 million phones due to a similar problem. But despite the risks, the flammable batteries continue to be used to power everything from smartphones and laptops to Teslas and Boeing 787s, simply because they are the smallest and lightest batteries available.

Experts assert the batteries are safe for the most part. They only have the potential to catch fire if they are charged too quickly and generate excess heat or have a manufacturing error that creates a short circuit. In the case of the hoverboards and Samsung phones, the explosions were caused by a combination of too many batteries crammed into a small space and a sub-optimized manufacturing process that put negative and positive poles into contact.
On January 13, a team of Stanford researchers announced that they had devised a way to stop the chemical reaction that causes Li-ion batteries to combust by using flame-retardant triphenyl phosphate (TPP). This is not the first time scientists have suggested using the chemical compound as a fire extinguisher. However, previous attempts to incorporate TPP resulted in reducing the battery’s performance making it impractical to use.
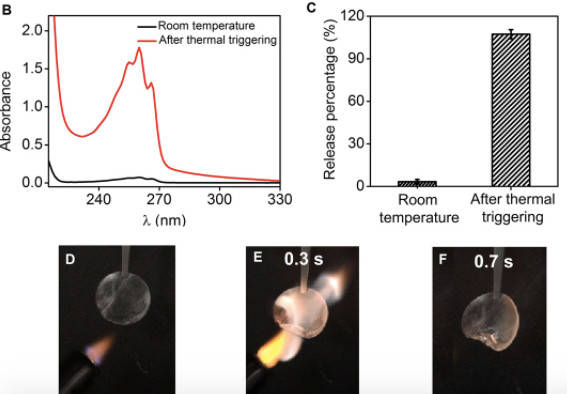
To overcome this issue, the Stanford team, led by postdoctoral research fellow Kai Liu, created a shell that acts as a barrier between the TPP and the Li-ion cell when the battery is working normally. But should the battery start to overheat, the shell melts, allowing the TPP to suppress the chemical reaction before it catches fire. In laboratory tests, the flame retardant was able to curb the overheating in just 0.4 seconds.
Even more importantly, only a small amount of TPP is needed to do the task, which means that the size and weight of the battery will not be affected. The fact that the fumes released are non-toxic is the icing on the cake. What is unclear, is if the battery or perhaps even the device needs to be replaced once the chemical compound is released. But given the alternative, we would rather spend money on a new battery, or even a new phone, wouldn’t you?
Resources: BBC.co.uk, theregister.co.uk,cnet.com,npr.org